Context
In this case study, we aim to present an example of how behavioural science methods can be used to help identify how to reduce antibiotic prescribing behaviour but crucially without creating a culture of blame which is often thought inevitable. In this context, culture of blame refers to an environment where individuals (such as general practitioners, or GP’s) are placed at fault for antibiotic prescribing (Locke, 2009). It is based on reviewing theories and methods of research in different social science disciplines and identifying an issue as potentially contentious and itself presenting a barrier for closer integrated and interdisciplinary work.
The use of behavioural science to address what has become seen as unnecessary and potentially damaging prescribing of antibiotics, especially in a primary care setting, commonly involves the inevitable blaming of individuals for implied bad behaviour (Khatri, Brown and Hicks, 2009). Interventions are often directed at educating them about the dangers of AMR to scare them out of such practice or moulding their motivations to cultivate better habits. The aim of this case study is to show how carefully designed and interdisciplinary social interventions can address structural barriers to behaving in ways the prescribers already think is good practice, rather than pointing out problematic motivations of poor practice.
Behavioural science methods and interventions to reduce antibiotic prescribing
The focus of this case study is a GP in a local practice in London, United Kingdom. She prescribes antibiotics for a patient with a nasty chest infection when she is unsure of the cause of infection. In this case, a behavioural scientist is working to influence how GPs prescribe antibiotics, and she is charged with co-designing interventions to address different ‘barriers’ to behaviour deemed desirable to reduce antibiotic prescribing. She is using the COM-B framework for helping to understand that behaviour.
Behavioural science methods
The COM-B framework (Figure 1) emphasises three factors in relation to behaviour. Capability refers to the physical or psychological ability to enact the behaviour. Motivation being the mechanisms that inhibit or activate behaviour. And opportunity being the physical and social environments that enable this behaviour.
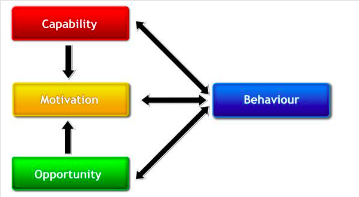
Figure 1: A visual representation of the COM-B framework as shown in (Michie, van Stralen and West, 2011). News media interest 1990-2019 (source: Davis et al., 2020b)
The behavioural scientist knows that information and motivation are components of the COM-B Framework and she has done a review of the existing literature which finds that educational and motivational interventions are most effective when accompanied with active interventions. Such interventions must also be multifaceted in nature to significantly influence long term changed behaviour (Roque et al., 2014).
She also knows that GPs probably already have some expertise in antimicrobial resistance though their professional training, journal articles, and conferences, and are likely to think reducing use of antibiotics is generally a good idea, having seen or read about patients with antibiotic resistant infections. So, she looks at the remaining components of the COM-B framework, namely capacity and opportunity. These will aid in addressing the structural/societal issues related to their prescribing behaviour.
On reading the results of her semi-structured survey, the behavioural scientist sees that the GP in question knows that the patient is a single father of three boys and is self-employed needing to get back to work as soon as possible to pay the bills. She learns further that he is not sleeping because of the chest infection, and that his elderly mother is moving into residential care 300 miles away at the weekend.
Any standard laboratory test results will currently take at least 72 hours. The GP understandably feels conflicted in her duties of care knowing that antimicrobial resistance is a growing problem. Yet, for the patient, as it stands, it looks like a course of antibiotics could help clear up a bacterial infection – if indeed there is one – and clear it up more quickly than leaving it to get better on its own. However, if the cause is viral, there is potential harm to the patient’s overall health and the antibiotic use could contribute to general resistance of microbes to it. The patient has tried over the counter remedies for his symptoms and is clearly struggling.
During the co-design workshop based on her results, the behavioural scientist is reassured that there is no need to try to change the GPs’ knowledge, motivation, or strength of will. Indeed, in discussion of all the options, the GPs confirm that they should not be held accountable for some of the difficult situations in which they must be the gatekeepers and stewards of antibiotics. The GPs at times also feel pressured to meet the patients expectations and reduce risk of other adverse health outcomes (Lopez-Vazquez, Vazquez-Lago and Figueiras, 2012). Some interventions could, they acknowledge, be perceived as blaming certain individual GPs especially if surveillance projects of GP prescribing habits is presented in isolation of context and is not anonymised but picked up as a problem for social sanction at best, and professional reprimand at worst.
Behavioural science intervention
The behavioural scientist persuades the GP practice to introduce one intervention which involves an extra service to increase the opportunities for GPs to reduce prescribing rates. This allows for a responsibility culture rather than a blame culture (Parker and Davies, 2020). These new services support patients when they are ill with extra and free childcare provision provided by local childminder services. She works with the local council to introduce a voucher system that allows for patients to receive vouchers from the GP that can be redeemed for the required number of free childminder days. This reinforces to the community the necessity of supporting one another.
She is also campaigning for more councils to introduce this system to help compensate certain patients who are refused antibiotics for lost work while they recover. These are structural provisions to supporting those patients who are already socially and economically disadvantaged to contribute to a collective effort to address antimicrobial resistance. Patients are very pleased to see the new provisions, which are considered newsworthy in the local Gazette, and readily join the campaign to garner support for patients in financial difficulty during periods of illness.
Given the administrative delays with sending samples to a centralised laboratory service and the difficulties GPs report in her survey about reconciling apparently conflicting duties, the behavioural scientist collaborates with health system academics to find a feasible way to decentralise laboratory services. They are able to introduce more efficient existing testing methods that reduce the delay in sample processing. GPs are keen to participate and find further efficiencies for the practice which result from these technologies with fewer patient visits and fewer laboratory tests.
Conclusion
Far from thinking of individual prescribers as being irresponsible or even reckless over their supposed stewardship of antibiotics, attending to certain structural issues can further be seen to redress known social and health inequalities.
Points of discussion
Pedagogical notes
PhD student and teaching assistant at University College London
Professor of bioethics at University College London